Unexpected Mutations after CRISPR in vivo editing – post-commentary
You might have heard or participated in the global discussion over the recently published Nature Commentary that described >1000 off-target mutations in CRISPR-edited mice.
The paper reported a small study involving three mice but gained enough virality online to trigger a significant drop in share prices of companies founded based on CRISPR gene-editing – Editas Medicine, CRISPR Therapeutics and Intellia Therapeutics.
Here is a summary of the study, with respective concerns raised by the scientific community regarding the validity of the findings. These are highlighted *in blue with further explanations below:
- FVB/NJ mice were used in the study.These mice are a highly inbred strain (F87 on Dec 2002) originating from the NIH but transferred to The Jackson Laboratory for maintenance and sale. They are homozygous for the Pde6brd1 allele, subjecting them to early onset retinal degeneration.
- The same authors previously published a pretty decent paper where they functionally characterized a rescue of the retinal degeneration by correcting what was thought to be a nonsense mutation (Y347X, C>A) at exon7 of the Pde6β subunit. The same “rescued” mice, edited by CRISPR (F03 and F05), along with the control co-housed mouse that did not undergo editing, were used in this subsequent sequencing study. *Concern 1
- The CRISPR mutation was performed by introducing the sgRNA via a pX335 plasmid (which would co-express Cas9D10A nickase) into FVB/NJ zygotes, alongside a single-stranded oligo which acts as a donor to introduce a controlled mutation at the Pde6b. WT Cas9 protein was also introduced. *Concern 2
- DNA was isolated from spleen of the mice and whole genome sequencing was performed with an Illumina HiSeq 2500 sequencer with a 50X coverage for CRISPR-treated mice and 30X coverage for the control mouse.
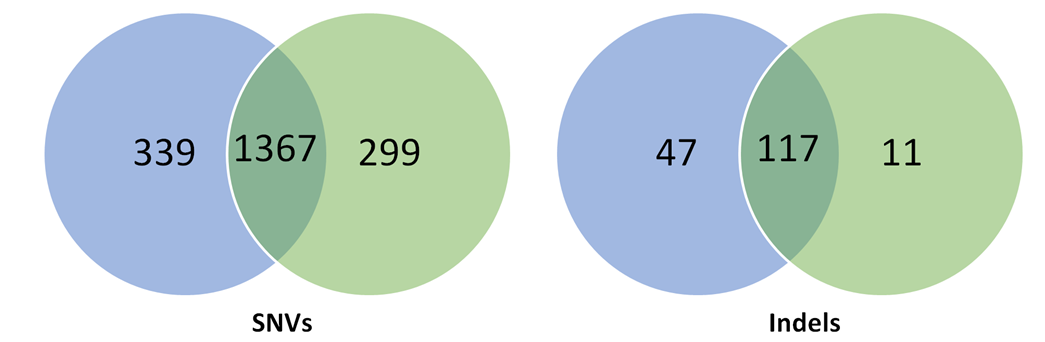
- The authors used three different algorithms to detect variants – Mutect, Lofreq and Strelka. The number of single nucleotide variants (SNVs) and insertion deletions (indels) detected that were absent in the control mouse are shown below for the two CRISPR-edited mice.
Overlap of SNV/indels detected in two CRISPR-edited mice – F03 mouse (blue), F05 mouse (green).
- Each of the variants were filtered against the FVB/NJ genome in the mouse dbSNP database (v138) and also against 36 other mouse strains from the Mouse Genome Project (v3). As none of the variants detected were found in these database genomes, the authors concluded they had to arise through CRISPR-editing. *Concern 3
- Interestingly, the top 50 predicted off-target sites showed no mutations. And in sites where mutations were detected, there was no significant sequence homology against the sgRNA used. The authors conclude in silico modelling fails to predict off-target sites. *Concern 4.
A number of criticisms have been raised regarding the study and the four main concerns highlighted are explained below:
Concern 1: The study only involved three mice, hence is too underpowered to draw any statistically significant conclusions. Further, the choice of control mouse simply being a co-housed mouse (no mention of its background) may fail to capture any genetic alterations induced by the experimental procedure or by genetic drift within a colony.
More appropriate controls may have included a mouse produced with a sham-injected zygote, a mouse where only Cas9 was introduced without an sgRNA, and a mouse with only sgRNA and ssDNA donor.
Parent mice should also have been sequenced to check if variants detected were already in the existing strain.
Concern 2: Cas9 was introduced both as a protein and in a plasmid. Talk about overkill! Though the plasmid form of Cas9 is the nickase version, where 2 sgRNAs are required to produce a double-strand break, having high levels of active Cas9 floating about has been demonstrated to increase the incidence of off-target effects.
Concern 3: Even though the authors filtered the variants found against mouse genome databases, this may not be sufficient to capture the extent of genetic drift that occurs over multiple generations of in-breeding.
Gaetan Burgio wrote that from his experience, the reference genomes found in databases often fail to capture the amount of variants that are specific to every breeding facility. Often large numbers of reference mice (1oo mouse exomes from > 50 founders) have to be sequenced to determine if SNPs were specific to the mouse strain and not induced by the test condition.
Editas and George Church’s group from Harvard also highlighted the high amount of overlap in SNVs/indels between the two CRISPR-edited mice which..
“strongly suggests the vast majority of these mutations were present in the animals of origin. The odds of the exact nucleotide changes occurring in the exact same position of the exact same gene at the exact same ratios in almost every case are effectively zero.”
Concern 4: Apart from the flaw that only one sgRNA was studied, Church’s group also claim the sgRNA studied had a high off-target profile. This sgRNA would apparently have failed their criteria for use as a therapeutic candidate. The table below shows the number of predicted off-target sites when allowing for 1-3 mismatches from the sgRNA sequence.
| Predicted off-target profile of sgRNA used in study | |
| Off-target sites with 1 mismatch | 1 |
| Off-target sites with 2 mismatches | 1 |
| Off-target sites with 3 mismatches | 24 |
What was surprising from the study however, was that despite the high off-targeting potential, mutations were not seen at predicted off-target sites.
The consensus therefore, by both Church’s group and the authors of the study was that one cannot rely on in silico prediction alone to account for off-target effects.
Calls are now being made to validate the study using the appropriate controls, or to compare the variants obtained with other more updated mouse genome SNP databases. I expect we will not hear the last of this study.
The study however, does re-enforce our message in a previous blogpost of validating CRISPR experiments with other techniques to establish gene function. It also highlights the extensive genetic heterogeneity seen now not only between cell lines, but between mouse strains. As always we recommend not being swept up in the hype, but to remain scientifically skeptical.
Want to receive regular blog updates? Sign up for our siTOOLs Newsletter: