Clearly compensating
Genetic compensation by transcriptional adaptation is a process whereby knocking out a gene (e.g by CRISPR or TALEN) results in the deregulation of genes that make up for the loss of gene function.
A 2015 study by Rossi et al. (discussed previously) alerted researchers that CRISPR/TALEN knock-out experiments may be subject to such effects.
Genetic adaption or compensation had been well known to mouse researchers creating knock-out lines. In fact, one of our company founders also ran into this when trying to confirm an RNAi phenotype in a knock-out mouse line. The knock-out mice, though not completely healthy, did not confirm the RNAi phenotype.
A paper published a couple years before the Rossi paper also showed clearly that knock-outs can create off-target effects via transcriptional adaptation.
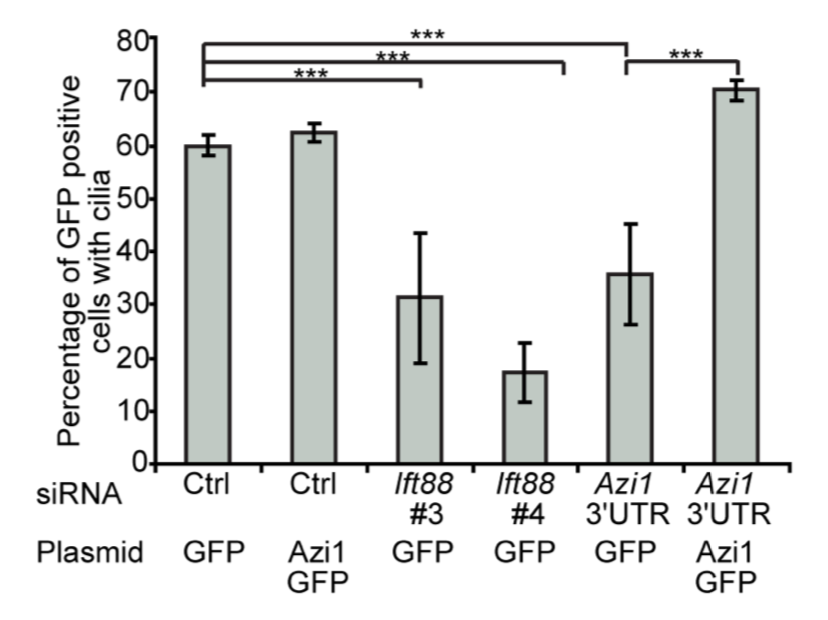
Hall et al. showed with an siRNA screen that the centrosomal protein Azi1 was required for ciliogenesis in mouse fibroblasts, confirming previous work in zebrafish and fly.
Their Azi1 siRNA targeted the 3′ UTR, and they were able to rescue the phenotype with a plasmid expressing just the CDS (bar at far right), confirming that their phenotype was due to on-target knockdown:
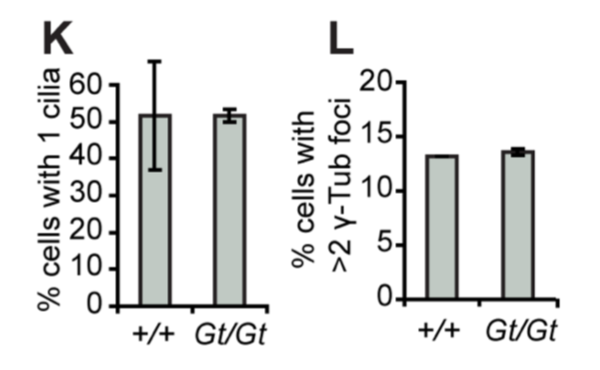
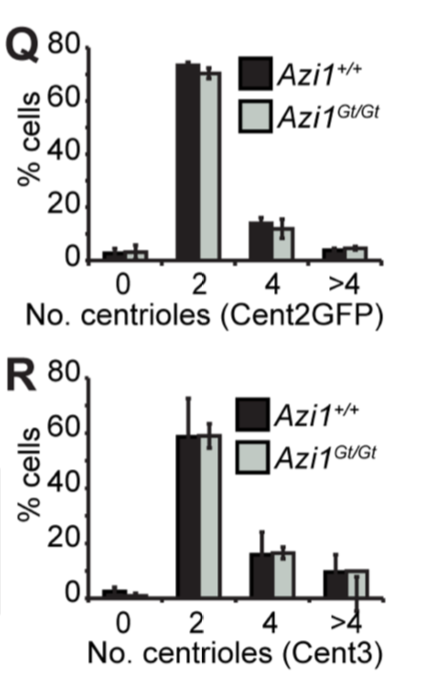
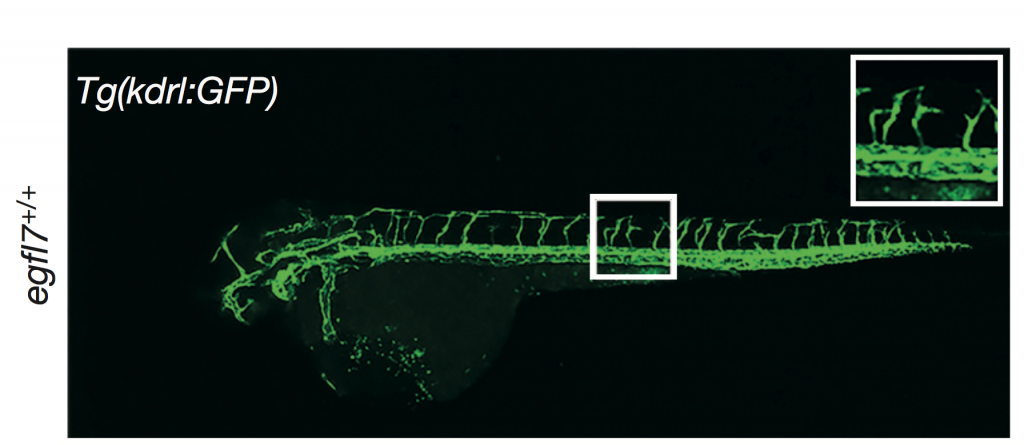
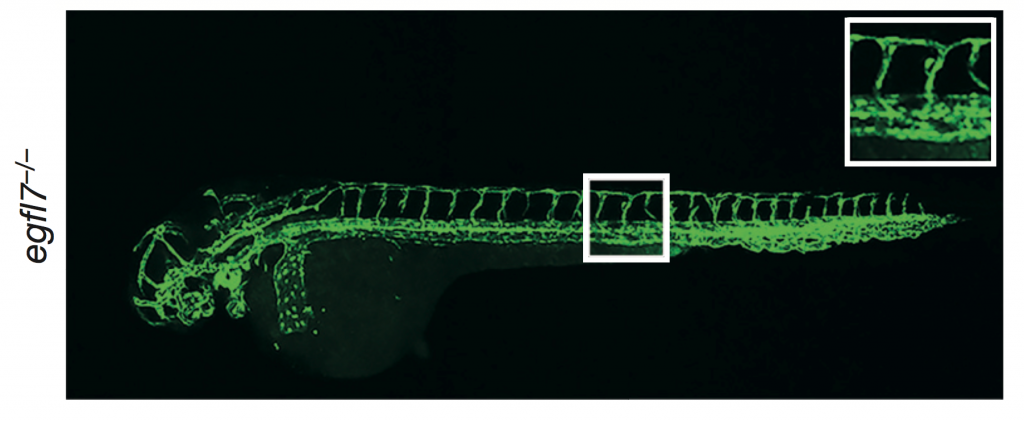
However, knock-out mouse embryonic fibroblast cells (created by gene trapping) did not show any differences in in the number of cilia, centrosomes, or centrioles compared to wildtype (+/+ is wild type, Gt/Gt is the homozygous knock-out):
The one phenotypic difference they observed was that male knock-out mice were infertile, due to defective formation of sperm flagella. Female mice had normal fertility. Both were compensating, but only one showed a visible phenotype.
The authors note the benefits of RNAi in comparison to knock-out screening:
Discrepancies between the phenotypic severity observed with siRNA knock-down versus genetic deletion has previously been attributed to the acute nature of knock-down, allowing less time for compensation to occur
The excitement surrounding CRISPR should not diminish the continued value of RNAi screening.